|
'Portrait of Joseph Roulin' (1888)
Boston, Museum of Fine Arts In the winter of 1888, Vincent van Gogh painted the portrait of his good friend and neighbor, Joseph Roulin who was born on 4 April 1841 in Lambesc located in southern France; he was a 47 year old Postman.
|
Vincent Willem van Gogh
|
'Worn Out: At Eternity's Gate' (1890)
Though he believed Van Gogh was suffering from a form of epilepsy caused by too much coffee and alcohol and too little food, his doctor, Dr. Rey never made an official diagnosis.
("Lust for Life", captures Vincent's depression). |
Gut bacteria and mind control: to fix your brain, fix your gut!
|
Microbes in your gut impact your brain, how people think, how people, and how your immune system functions. Colitis is an inflammation of the colon. Colitis may be acute and self-limited or long-term. It broadly fits into the category of digestive diseases. Colitis is the primary symptom before the onset of Autoimmune Diseases. (1), (2), (video)
In terms of cell numbers, we are about 10% human and 90% bacterial.
Xu, J., and J. I. Gordon. 2003. Honor thy symbionts. Proc. Natl. Acad. Sci. USA 100:10452-10459. [PMC free article] [PubMed] [Google Scholar] |
|
Some Diseases That Can Start with Your Gut Bacteria
|
Mental illness
By introducing healthier strains of bacteria, researchers have been able to treat behavior like depression, anxiety, and even autism
Colon cancer
Scientists have found that toxins produced by two types of bacteria—E. coli and B. fragilis—combine in the colon to damage DNA
|
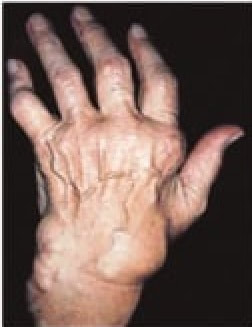
Rheumatoid arthritis
RA patients are more likely than healthy people to have several rare species of bacteria in their guts. In the a study, researchers transplanted a healthy strain of bacteria into mice with RA, and their symptoms improved. While the researchers aren’t sure why this approach works, they do believe the microbiome can stimulate the immune system to attack the body.
|
C. difficile
infection by C. difficile, a bacteria that causes diarrhea, stomach cramps, fever, and in severe cases, kidney failure. It often develops in people who have taken heavy-duty antibiotics that killed off the normal bacteria in their digestive tract allowing opportunistic bacteria an opportunity to flourish. Today Fecal transplants have a 90 percent success rate.
|
Multiple sclerosis
A 2017 study found that two types of bacteria are more common in MS patients, and the strains can cause cellular changes in healthy blood, increasing the likelihood of a dangerous autoimmune response, according to STAT News.
Diabetes
dietary fiber helps by nourishing gut bacteria that help influence insulin
|
Irritable bowel syndrome (IBS)
Fasting. In this study, researchers looked at fifty-eight patients with IBS and divided them into two groups. One group was given prescription meds and psychotherapy to reduce IBS symptoms, while the other group fasted for 10 days, drinking only water. The fasting group reported improvement in 7 of 10 symptoms.
|
|
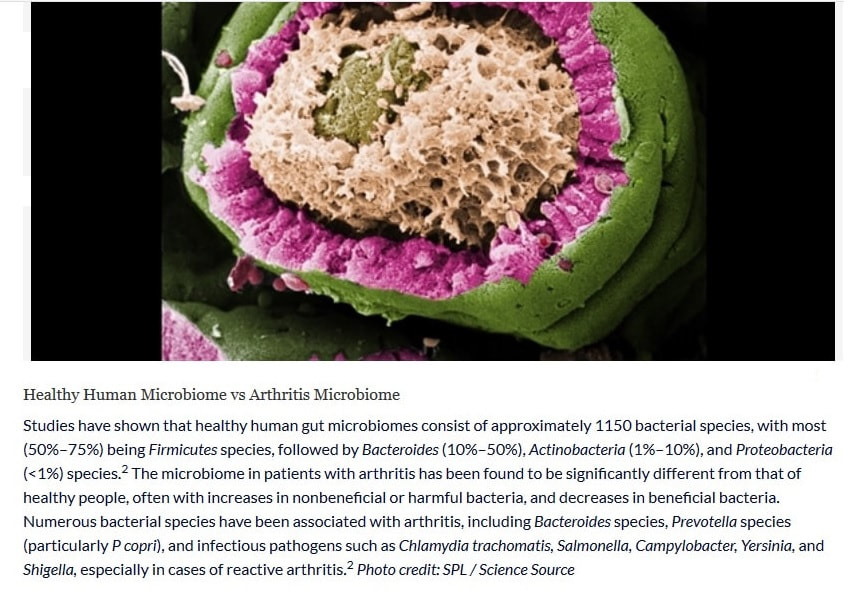
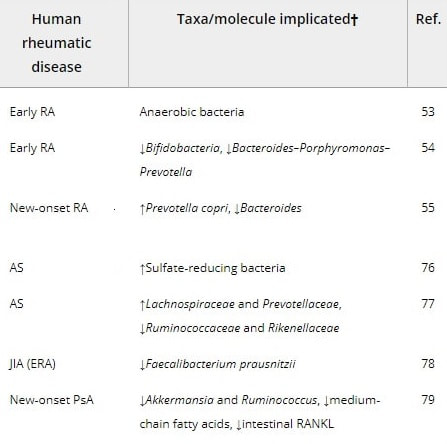
The alteration in the composition and function of intestinal microbiome has been associated with rheumatoid arthritis, that is patients having decreased gut microbiome diversity as compared to the healthy individuals.[37] Smoking is also one of the causative factors of rheumatoid arthritis as it contributes to the development of RF factor which is up to three times more common in smokers than non-smokers.[38,39] The initiation of RA
|
The initiation of RA has been associated with mineral oils, silica exposure, diet factors and blood transfusion. The probable causative factors are excessive intake of saline and uncooked food, and intake of burning sensation producing food during digestion (Table 1).
Source: Medicinal plants used against various inflammatory biomarkers for the management of rheumatoid arthritis S Singh, TG Singh, K Mahajan… - Journal of Pharmacy …, 2020 - Wiley Online Library https://onlinelibrary.wiley.com/doi/pdf/10.1111/jphp.13326 |
Based on these data, authors have proposed that
Proteobacteria may represent a “microbial signature” of disease.
“Proteobacteria: microbial signature of dysbiosis in gut microbiota,” Trends in Biotechnology, vol. 33, no. 9, pp. 496–503, 2015.
- The PCB-polluted soil exhibited a high bacterial diversity which included representatives of two novel lineages. The apparent abundance of bacteria affiliated to the beta-subclass of the Proteobacteria, and to the genus Burkholderia in particular, was confirmed by fluorescence in situ hybridization analysis.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC92809/
- Proteobacteria: A Common Factor in Human Diseaseshttps://www.hindawi.com/journals/bmri/2017/9351507/
Microbiota represents the entire microbial community present in the gut host. It serves several functions establishing a mutualistic relation with the host. Latest years have seen a burst in the number of studies focusing on this topic, in particular on intestinal diseases. - In this scenario, Proteobacteria are one of the most abundant phyla, comprising several known human pathogens.
Images for Proteobacteria, arthritis
|
What bacteria causes rheumatoid arthritis?
Clinical and animal model studies have suggested that infections by many microorganisms, such as Porphyromonas gingivalis (P. gingivalis), Proteus mirabilis (P. mirabilis), Epstein–Barr virus (EBV), and mycoplasma contribute to the etiopathogenesis of RA (Table 1). Common RA-associated microbes. Reactive arthritis - Chlamydia trachomatis, Salmonella, Yersinia, Shigella
Role of Gut Microbiome on Spondyloarthritis
https://www.infectiousdiseaseadvisor.com/slideshow/gi-illness/role-of-gut-microbiome-on-spondyloarthritis/ |
|
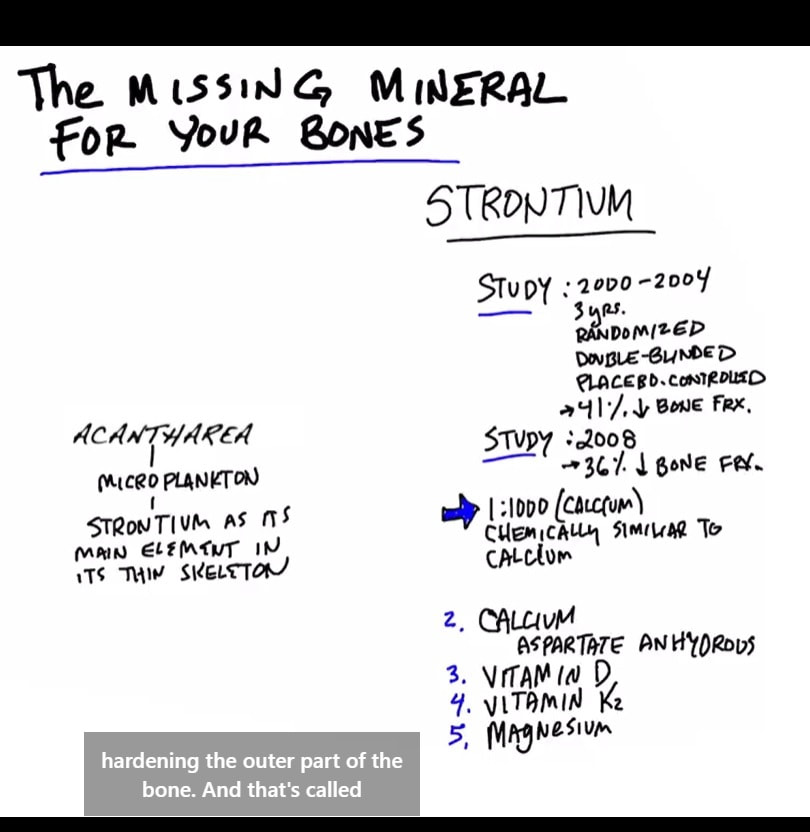
Mineral for strengthening your bones
WONDER WHY THE PALMS OF YOUR HANDS ARE SO RED. . Dr. Eric Berg October 23 2021 9 Signs of Toxin Build-up in the Liver Today we’re going to talk about the nine things your skin can tell you about your liver. https://www.facebook.com/drericberg/videos/1205403849944126 ALT: Seven Remedies for an Inflamed Liver https://www.facebook.com/watch/?v=1205403849944126 The Acantharea (Acantharia) are a group of radiolarian[1] protozoa, distinguished mainly by their strontium sulfate skeletons. Acantharians are heterotrophic marine microplankton that range in size from about 200 microns in diameter up to several millimeters. Some acantharians have photosynthetic endosymbionts and hence are considered mixotrophs. |
Bacteroides species
Five classes of proteobacteria (all are gram negative)
1. Alphaproteobacteria, 2. Betaproteobacteria, 3. Gammaproteobacteria, 4. Deltaproteobacteria, 5. Epsilonproteobacteria
1. Alphaproteobacteria, 2. Betaproteobacteria, 3. Gammaproteobacteria, 4. Deltaproteobacteria, 5. Epsilonproteobacteria
|
1. Alphaproteobacteria - all are oligotrophs, which is organisms capable of living in low-nutrient environments such as deep oceanic sediments, glacial ice, or deep undersurface soil.
|
Chlamydia (Alpha): STD; resistant to cellular defenses
Rickettsia. (Alpha): Tick formed- rocky Mountain spotted fever, - Obligate intracellular pathogen: require living host cells to grow and propagate.
2. Betaproteobacteria - are eutrophs (or copiotrophs), meaning that they require a copious amount of organic nutrients. areobic and anerobic
Neisseria (Beta) Gonorrhea and Meningitis; req. moisture and lost of CO2
Bordetella (Beta) whooping cough; produce toxins to paralyze lung cilia
3. Gammaproteobacteria
Pseudomonas aeruginosa (Gamma)
Infection of wounds; urinary and resp. Tract
Infection of wounds; urinary and resp. Tract
Haemophilus influenzae (Gamma) Agent for lower and upper resp. Infections
Pasteurlla haemolytia (Gamma) Severe pneumonia in animals
Legionalla pneumophila (Gamma) Legionnaires disease; water contamination
Enterbacter family (Gamma) Coliforms- ferment lactose, NonColiforms- does not ferment lactose
Vibrio (Gamma) Toxin can cause hypersecretion of water/electrolytes in G.I tract
4. Deltaproteobacteria
|
Myxobacterium (Delta)
Live in soil and can move by gliding |
Bdelloribrio (Delta)
Parasitic; infecting other bacteria |
Desulfovibrio (Delta)
Removal of toxic and radioactive waste |
5. Epsilonproteobacteria
|
Campylobacter (Epsilon)
food poisoning |
Heliobacter (Epsilon)
stomach ulcers; damage stomach lining |
Prevotella species (P copril)
Description and SignificancePrevotella sp. are among the most numerous microbes culturable from the rumen and hind gut of cattle and sheep, where they help the breakdown of protein and carbohydrate foods. They are also present in humans, where they can be opportunistic pathogens. Prevotella, credited interchangably with Bacteroides melaninogenicus, has been a problem for dentists for years. As a human pathogen known for creating periodontal and tooth problems, Prevotella has long been studied in order to counteract its pathogenesis (AAP).
Genome StructureTwo strains of Prevotella have been completely sequenced: Prevotella intermedia 17 and Prevotella ruminicola 23. Both strains were sequenced by TIGR. The sequencing of these two genomes has lead to increased ability to combat Prevotella strains' pathogenic abilities.
Genome StructureTwo strains of Prevotella have been completely sequenced: Prevotella intermedia 17 and Prevotella ruminicola 23. Both strains were sequenced by TIGR. The sequencing of these two genomes has lead to increased ability to combat Prevotella strains' pathogenic abilities.
IgA (dimer) : Antibodies that defend the body from bacterial infection
|
IgAs are immunoglobulins, or antibodies not typically found in the blood but in secretions of the gut, saliva, sweat, breast milk, respiratory tract, and urinary tract.
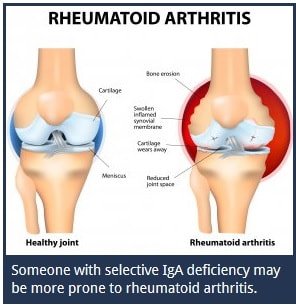
There are various health conditions that can lead to low levels of Immunoglobulin A in the body. When the body does not have sufficient quantities of Type A Immunoglobin, the person may be diagnosed with selective IgA deficiency. This is considered a disease of the autoimmune system.
|
Lower fecal IgA in early life has been linked to future allergic conditions in infants born to mothers experiencing distress may have a greater risk of developing diseases of the immune system.
|
esearcher Liane Kang talks about her findings on pre and postnatal distress experienced by the mother that is linked to reduced Immunoglobulin A levels in infant feces (video). (1), (2)
Maternal distress during or after pregnancy is associated with changes in early life gut immunity in infants. Secretory immunoglobulin produced in the gut known as GA for short IgA is the main antibody for gut mucosal defense which is provided to infants via mother's breast milk which:
Some diseases, such as gonorrhea, can destroy Immunoglobulin A in the body, leading to a deficiency.
|
American Gut Project
|
What's In Your Gut?
|
Explore illness and symptoms caused by bacteria (or lack of) through this on-line interactive course. Top rated Instructors make this subject matter easy, fun, and fascinating to learn even if you don't have a science background. Near the completion of the course you will be given an opportunity to study your gut microbes by submitting your sample to the American Gut Project (U.S. $100). No harm in doing this before the course starts as it will give you an edge to understand what ails you or enhances your health.
|
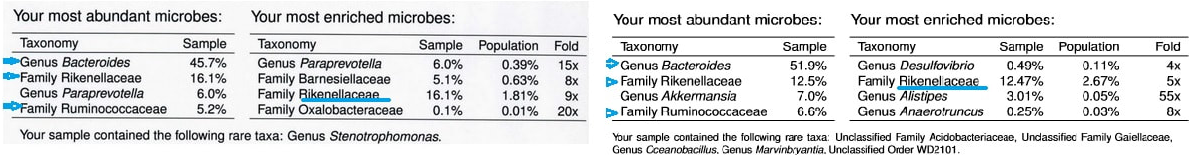
The American Gut Project has proven that micro-organisms play a role in not just Rheumatoid Arthritis but numerous other disease as well. Below are two examples of gut microbes test results from two females who have had Rheumatoid Arthritis for a number of years and have modified their diet in hopes to rid themselves of the disease, stop medications, and improve gut flora.
EXAMPLE of an American Gut 101 - Human Microbiome of a person with RA
Kingdom Phylum Class Order Family Genus Type species
Bacteria Bacteroidetes Bacteroidia Bacteroidales Bacteroidaceae Bacteroides ---
Bacteria Bacteroidetes Bacteroidia Bacteroidales Rikenellaceae --- ---
Bacteria Verrucomicrobia Verrucomicrobiae Verrucomicrobiales Verrucomicrobiaceae Akkermansia increased by Gut Inflammation and Mucus (1)
Bacteria Firmicutes Clostridia Clostridiales Ruminococcaceae --- ---
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae --- ---
Bacteria Bacteroidetes Bacteroidia Bacteroidales Rikenellaceae Alistipes ---
Bacteria Firmicutes Clostridia Clostridiales 2.32
Bacteria Firmicutes Clostridia Clostridiales Ruminococcaceae Ruminococcus ---
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae Dorea ---
Paraprevotelia
Barnesiellaceae
Oxalobacteraceae
Note: Where there are blanks in the table, the taxonomy could not be resolved in finer detail.
Note: The percentages listed represent the relative abundance of each taxon.
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae Dorea 1.21
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae Dorea 1.21
The soil of forested tundra was dominated by uncultivated members of the family Planctomycetaceae (53–71% of total Planctomycetes-like reads), while sequences affiliated with the Phycisphaera-related group WD2101 (recently assigned to the order Tepidisphaerales) were most abundant in peat (28–51% of total reads).(1)
Kingdom Phylum Class Order Family Genus Type species
Bacteria Bacteroidetes Bacteroidia Bacteroidales Bacteroidaceae Bacteroides ---
Bacteria Bacteroidetes Bacteroidia Bacteroidales Rikenellaceae --- ---
Bacteria Verrucomicrobia Verrucomicrobiae Verrucomicrobiales Verrucomicrobiaceae Akkermansia increased by Gut Inflammation and Mucus (1)
Bacteria Firmicutes Clostridia Clostridiales Ruminococcaceae --- ---
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae --- ---
Bacteria Bacteroidetes Bacteroidia Bacteroidales Rikenellaceae Alistipes ---
Bacteria Firmicutes Clostridia Clostridiales 2.32
Bacteria Firmicutes Clostridia Clostridiales Ruminococcaceae Ruminococcus ---
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae Dorea ---
Paraprevotelia
Barnesiellaceae
Oxalobacteraceae
Note: Where there are blanks in the table, the taxonomy could not be resolved in finer detail.
Note: The percentages listed represent the relative abundance of each taxon.
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae Dorea 1.21
Bacteria Firmicutes Clostridia Clostridiales Lachnospiraceae Dorea 1.21
The soil of forested tundra was dominated by uncultivated members of the family Planctomycetaceae (53–71% of total Planctomycetes-like reads), while sequences affiliated with the Phycisphaera-related group WD2101 (recently assigned to the order Tepidisphaerales) were most abundant in peat (28–51% of total reads).(1)
At Risk Populations for Rheumatoid Arthritis. Opens in a new tab
In this research proposal, we intend to study the role intestinal bacteria and bacteria on other body surfaces, such as the skin, in the development of inflammatory arthritides, including rheumatoid arthritis (RA) and with a concentrated effort to recruit from an at risk population. Given the fact that RA concordance in MZ twins is =15%, we hypothesize that among individuals with similar genetic background, environmental and/or microbiome factors are predictive for RA development, or protective in healthy, at risk populations. Samples of blood, urine, skin swab and stool will be assessed to establish clinical, immunologic and microbiome differences among populations at risk for RA and other related autoimmune diseases, including, but not limited to: relatives of patients diagnosed with RA (including twins), psoriatic arthritis (PsA), multiple sclerosis (MS), ankylosing spondylitis (AS); and individuals with skin psoriasis (but no arthritis), periodontal disease, as well as cigarette smokers. Individuals with a confirmed diagnosis of skin psoriasis (but not arthritis) will be considered ‘at risk’ for the development of Psoriatic Arthritis and be the sole family member to be enrolled and followed at six month intervals over a five year period.
https://clinicaltrials.med.nyu.edu/clinicaltrial/1018/at-risk-populations-rheumatoid/
In this research proposal, we intend to study the role intestinal bacteria and bacteria on other body surfaces, such as the skin, in the development of inflammatory arthritides, including rheumatoid arthritis (RA) and with a concentrated effort to recruit from an at risk population. Given the fact that RA concordance in MZ twins is =15%, we hypothesize that among individuals with similar genetic background, environmental and/or microbiome factors are predictive for RA development, or protective in healthy, at risk populations. Samples of blood, urine, skin swab and stool will be assessed to establish clinical, immunologic and microbiome differences among populations at risk for RA and other related autoimmune diseases, including, but not limited to: relatives of patients diagnosed with RA (including twins), psoriatic arthritis (PsA), multiple sclerosis (MS), ankylosing spondylitis (AS); and individuals with skin psoriasis (but no arthritis), periodontal disease, as well as cigarette smokers. Individuals with a confirmed diagnosis of skin psoriasis (but not arthritis) will be considered ‘at risk’ for the development of Psoriatic Arthritis and be the sole family member to be enrolled and followed at six month intervals over a five year period.
https://clinicaltrials.med.nyu.edu/clinicaltrial/1018/at-risk-populations-rheumatoid/
|
Bacteroides
Bacteria commonly found in yogurt, Lactobacillus casei and Lactobacillus acidophilus, reduced joint inflammation more effectively than the NSAID indomethacin.(1)
Prevotella copri
In accordance, genome analysis of Prevotella copri showed it was deficient in the ability to degrade host glycans and is more genetically equipped for plant glycan degradation.[2] SoMAS - Recognition of "self" and "non-self" Glycans by Carbohydrate-binding Proteins (3)
We identified the presence of Prevotella copri as strongly correlated with disease in new-onset untreated rheumatoid arthritis (NORA) patients. Increases in Prevotella abundance correlated with a reduction in Bacteroides and a loss of reportedly beneficial microbes in NORA subjects. We also identified unique Prevotella genes that correlated with disease. Further, colonization of mice revealed the ability of P. copri to dominate the intestinal microbiota and resulted in an increased sensitivity to chemically induced colitis. This work identifies a potential role for P. copri in the pathogenesis of RA.(4) The immune response to Prevotella bacteria in chronic inflammatory disease. Larsen JM Immunology. 2017 Aug;151(4):363-374. Epub 2017 Jun 20. Prevotella abundance is reduced within the lung microbiota of patients with asthma and chronic obstructive pulmonary disease. Increased Prevotella abundance is associated with augmented T helper type 17 (Th17) -mediated mucosal inflammation, which is in line with the marked capacity of Prevotella in driving Th17 immune responses in vitro. Studies indicate that Prevotella predominantly activate Toll-like receptor 2, leading to production of Th17-polarizing cytokines by antigen-presenting cells, including interleukin-23 (IL-23) and IL-1. Furthermore, Prevotella stimulate epithelial cells to produce IL-8, IL-6 and CCL20, which can promote mucosal Th17 immune responses and neutrophil recruitment. Prevotella-mediated mucosal inflammation leads to systemic dissemination of inflammatory mediators, bacteria and bacterial products, which in turn may affect systemic disease outcomes. Studies in mice support a causal role of Prevotella as colonization experiments promote clinical and inflammatory features of human disease. When compared with strict commensal bacteria, Prevotella exhibit increased inflammatory properties, as demonstrated by augmented release of inflammatory mediators from immune cells and various stromal cells. These findings indicate that some Prevotella strains may be clinically important pathobionts that can participate in human disease by promoting chronic inflammation. Rikenellaceae - researchers reported that extroverted personality traits were associated with the abundances of microbes from the Rikenellaceae and
Ruminococcaceae families (1) Frequency of HLA-B27 alleles in Brazilian patients with Psoriatic Arthritis. http://www.science.gov/topicpages/h/hla-b27+positive+patients.html Rikenellaceae bacterium 4-1-11 Rikenellaceae bacterium DTU049) Ruminococcaceae |
Bacteroides and sphingolipids (Bacteroides spp)
- Role of Bacteroides - Signaling host to help enable persistence
- Bacteroides spp. - Excess of this in gut may underlie autoimmune diseases (multiple sclerosis)
- Make unusual lipids - Use sphingosine, not glycerol in lipid backbone
- Sphingolipids common in mammalian tissues (gangliosides, sphingomyelin)
- Role of Bacteroides - Signaling host to help enable persistence
- Bacteroides spp. - Excess of this in gut may underlie autoimmune diseases (multiple sclerosis)
- Make unusual lipids - Use sphingosine, not glycerol in lipid backbone
- Sphingolipids common in mammalian tissues (gangliosides, sphingomyelin)
Akkermansia muciniphila
the only currently known species within genus Akkermansia, can reside in the human intestinal tract and is currently being studied for its effects on human metabolism. Recently performed studies in rodents have indicated that Akkermansia muciniphila in the intestinal tract may mediate obesity, diabetes, and inflammation.[6][7][8][9][*]
Videeo: Suzanne Humphries, MD, speaking on Polio at the Association of Natural Health Conference
Videeo: Suzanne Humphries, MD, speaking on Polio at the Association of Natural Health Conference
Rikenellaceae
Frequency of HLA-B27 alleles in Brazilian patients with Psoriatic Arthritis. http://www.science.gov/topicpages/h/hla-b27+positive+patients.html
Rikenellaceae bacterium 4-1-11, Rikenellaceae bacterium DTU049)
Rikenellaceae bacterium 4-1-11, Rikenellaceae bacterium DTU049)
Dialister, Rikenellaceae, Ruminococcaceae, and Parabacteroides
How micro-organisms play a role in rheumatoid arthritis
|
capable of initiating a harmful immune response in individuals constitutionally predisposed to rheumatoid arthritis.
(Dr Alan G S Hill, 1968 ) |
|
Diphtheroids
|
- P. copri has been correlated with onset of rheumatoid arthritis.[12]
- Llinks between dysbiosis and rheumatoid arthritis, (Björkstén, 1999, Penders et al., 2007, Vaahtovuo et al., 2008).
- Elinav E, Strowig T et al. (2011) cell, study indicates that deficiencies in the NLRP6 pathway should be added to the list of host genetic factors that may drive disease-specific alterations in the microbiota, which in turn may promote disease in these hosts or in individuals who have been exposed to these microbial communities and who have also experienced disruption in their gut epithelial barrier function due to a variety of insults.
- Fecal microbiota in early rheumatoid arthritis
J. Rheumatol., 35 (2008), pp. 1500-1505
View Record in Scopus or Google Scholar
|
One thing people don’t realize is these intestinal bugs can wear down the lining in the intestines. When the lining has been worn down the toxins that are in the intestines start to get through and get into the blood stream. These toxins can then cause a variety of other problems.
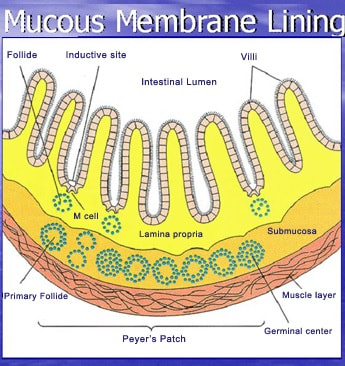
Abov is a picture of the lining of your intestines. You’ll see each yellow finger, called villi, have a whitish border. In your intestinal tract you make stool and stool or fecal material AKA poop is pretty toxic stuff. That border around the yellowish fingers (villi) in your intestines protect your body by stopping the toxins from getting into your blood stream. Damaged villus lay on its side and some of the fingers may be missing. Parasites, Yeast, Fungus, Mold and Bacteria, can all play a part in destroying the protective red cells inside the intestines. When the protective border gets destroyed or damaged the toxins get through to your blood stream. |
There are 132 different types of parasite and worms can survive in the human body.
Is it possible you have a parasite, yeast, bacteria or some other critter in your gut that shouldn’t be there? Yes! When a person has some type of bug in the intestines they can have several different signs or symptoms:
|
|
Bacteria, viruses, and parasites
Epithelial cells express ABH and Lewis antigens, which are effectively cell-surface glycoconjugates used by parasites, bacteria, and viruses as receptors for attachment, resulting in different susceptibilities depending on the antigen profile of an individual.11
By using the same blood group antigens as their host, certain microbial parasites utilize molecular mimicry as a defense against the host’s immune system.11 The chemical signatures on the membranes of many gram-negative organisms such as Escherichia coli resemble A and B blood group antigens; in vitro experiments have shown that anti-B antibodies kill E. coli, and anti-A and anti-B antibodies may therefore play a similar role in destroying gram-negative bacteria in vivo.14 |
Type O blood group is associated with increased incidence of
Type A blood group is associated with increased incidence of
Type B blood group is associated with increased incidence of
Type AB blood group is associated with increased incidence of
Non-secretors have an increased incidence of
References: 1) https://www.youtube.com/watch?v=-7RhiN5-2WY 11) Yamamoto F, Cid E, Yamamoto M, Blancher A. ABO research in the modern era of genomics. Transfus Med Rev. 2012;26:103–118. doi: 10.1016/j.tmrv.2011.08.002. [PubMed] [CrossRef] [Google Scholar] 14) Garratty G. Blood groups and disease: a historical perspective. Transfus Med Rev. 2000;14:291–301. doi: 10.1053/tmrv.2000.16228. [PubMed] [CrossRef] [Google Scholar] |
|
H. pyloriIn 1954, it was reported that the incidence of peptic ulcers (gastric and duodenal) was 20% higher for group O individuals than group A individuals, with the incidence of duodenal ulcers 35% higher in group O individuals than in group A, B, and AB individuals, and 50% higher in non-secretors (who make up 20% of the population).14 For group A and B non-secretors, the relative risk was 1:6; for group O secretors, the relative risk was 1:35; and for group O non-secretors, the relative risk was 2:5.14 Secretor status and H. pylori infection are independent and significant risk factors for gastroduodenal disease, with a relative risk of 1.9 for non-secretors vs secretors.25 Although gastritis and gastric ulcers are associated with H. pylori infection, more recent studies have reported that different strains of H. pylori showed varying preferences for each blood group antigen, and in fact, 95% of the strains
|
|
did not show a preference for blood group O antigens.11
Interestingly, although only 5% of the strains prefer the H antigen in
the general population, in those of American Indian heritage (a group
O-dominant population), 60% of the strains show this preference.11 READ MORE
Creating a dietary plan to feed the gut microbiome
We develop our microbiome in early life we are born with the very simple microbiome that we inherit from our mothers he arises in utero or is contributed to through the birthing process babies who are born through the vaginal canal have a preponderance of lactobacillus species which are the dominant organisms in the female vaginal tract babies who commit the Sun roof by cesarean section end up quite frequently with organisms we find on the skin Staphylococcus and streptococcus suggesting that very early life postnatal exposures influence those communities of microbes that are found in the very early gut
Microbial overgrowth and increased inflammation from gut issues can change the body’s need for vitamin D. In someone that is having chronic infections; it is not odd to see their blood test results show that they have high 1-25 hydroxy and low 25-hydroxy. Elevated 1-25 hydroxy is one way the body tries to fight the overgrowth through chronic immune activation and to activate VDR receptors to enhance the immune system. Their VDR receptors become changed epigenetically, possibly from your chronic infections or environment. 1-25 vitamin D increases CD14 gene expression, which helps the immune system recognize endotoxins and eliminate them. The less active your VDR receptors are in the gut, the more vulnerable you are to endotoxins and leaky gut. “Autoimmune” conditions that improve once vitamin D is normalized in the body and gut health is restored include: •Rheumatoid arthritis, Alzheimer’s, Diabetes (type 1 and 2), IBD, Multiple Sclerosis, Parkinson’s disease, Psoriasis, Systemic lupus erythematosus
Always make sure the vitamin D in the supplement is D3 and not the inferior D2. It is always best to get your vitamin D from proper sunlight exposure instead of supplements.
Microbial overgrowth and increased inflammation from gut issues can change the body’s need for vitamin D. In someone that is having chronic infections; it is not odd to see their blood test results show that they have high 1-25 hydroxy and low 25-hydroxy. Elevated 1-25 hydroxy is one way the body tries to fight the overgrowth through chronic immune activation and to activate VDR receptors to enhance the immune system. Their VDR receptors become changed epigenetically, possibly from your chronic infections or environment. 1-25 vitamin D increases CD14 gene expression, which helps the immune system recognize endotoxins and eliminate them. The less active your VDR receptors are in the gut, the more vulnerable you are to endotoxins and leaky gut. “Autoimmune” conditions that improve once vitamin D is normalized in the body and gut health is restored include: •Rheumatoid arthritis, Alzheimer’s, Diabetes (type 1 and 2), IBD, Multiple Sclerosis, Parkinson’s disease, Psoriasis, Systemic lupus erythematosus
Always make sure the vitamin D in the supplement is D3 and not the inferior D2. It is always best to get your vitamin D from proper sunlight exposure instead of supplements.
Our largest collection of microorganisms resides in the intestine
General features of the human intestinal microbiota
General features of the human intestinal microbiota
|
Second, population density increases by ≈8 orders of magnitude from the proximal small intestine (103 organisms per milliliter luminal contents) to the colon (1011 per g of contents) (5). Biodiversity also appears to increase along this axis, although the extent of diversification has yet to be systematically defined with secure region-specific sampling methods and enumeration methods that do not require cultivation (e.g., sequencing libraries of 16S rDNA amplicons).
|
Third, the microbiota functions as a multifunctional organ whose component cell lineages provide metabolic traits that we have not fully evolved in our own genome. These traits include the ability to break down otherwise indigestable plant polysaccharides (16, 17), biotransformation of conjugated bile acids (18), degradation of dietary oxalates (19), and synthesis of certain vitamins (20).
|
Fourth, postnatal colonization of our intestine educates our immune system, so we become tolerant of a wide variety of microbial immunodeterminants.
This education appears to reduce allergic responses to food or environmental antigens (21). |
Bifidobacterium
Autoimmune disorders occupy a prominent position among diseases that have long been thought to be triggered by micro-organisms. In particular, accumulating evidence suggests that the oral and intestinal microbiomes have a role in the development of rheumatoid arthritis (RA). The microbiome and rheumatoid arthritis link: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3275101/
Microbiome test results of the Hadza and/or Burkina Faso people have a completly different set of microbes than westerns. Bifidobacteria and Lactobacilli are not present in hadza children.
https://www.google.ca/search?q=biffobacteria+and+lactobacilia+are+not+present+in+hazda+children&ie=utf-8&oe=utf-8&gws_rd=cr&ei=wMfqV5azE-jNjwSyj4yQDg#q=bifidobacteria+and+lactobacilli+are+not+present+in+hadza+children
microbiome test results of the Hadza and/or Burkina Faso people
https://scholar.google.ca/scholar?hl=en&q=microbiome+test+results+of+the+Hadza+and%2For+Burkina+Faso+people&btnG=&as_sdt=1%2C5&as_sdtp=
|
Gut Microbes Linked to Rheumatoid Arthritis The researchers found that 75% of people with new-onset, untreated rheumatoid arthritis had the bacterium Prevotella copri in their intestinal microbiome. In comparison, it was present in 12% of people with chronic, treated rheumatoid arthritis, 38% of people with psoriatic arthritis, and 21% of those in the control group. Increased levels of P. copri correlated with reductions in several groups of beneficial microbes, such as Bacteroides. The researchers performed more complete DNA sequencing on a subset of samples and identified unique Prevotella genes that correlated with rheumatoid arthritis. (1) Asquith, Mark, Sternes, Peter R., Costello, Mary‐Ellen, Karstens, Lisa, Diamond, Sarah, Martin, Tammy M., et al. (2019) HLA alleles associated with risk of ankylosing spondylitis and rheumatoid arthritis influence the gut microbiome. Arthritis & Rheumatology, 71(10), pp. 1642-1650.
Viruses enter our bodies silently and invisibly and then parasitize our cells in a way that is almost totally beyond our control.
|
Sometimes it's the treatment that weakens your immune system, Chemotherapy or other immunosuppressive drugs such as methotrexate.
|
The gut is 80% of your Immune System
Rheumatoid arthritis patients have altered gut and oral bacteria [1].
Microbial diversity is significantly diminished in guts of patients suffering from Crohn’s disease [2].
By design, the cells which make up your gut's lining are resilent and constantly rebuilding themselves to fortify the barrier on a daily bases. This constant replacement takes a lot of energy and nutrients like B vitiams, amino acids, and zinc.
Gut-irritating foods (wheat, gluten), cell-inflaming foods (sugar), and nutrient-depleated foods (), set the stage for a inflammatory state. If the source of irritation and inflamation is constant it can lead to a condition known as leaky gut syndrome (LGS).
LGS also known as intestinal permeability, is associated with rheumatoid arthritis , diabetes, crohn's, and the list goes on. When the intestinal cell wall weakens, gaps in the barrier form which allow toxic chemicals, pathogenic viruses, parasites, and bacteria to slip through. The immune system tries to combat these foreign invaders by using chemicals which cause damage to the nearby intestinal lining.
Akkermansia muciniphila is a species of human intestinal mucin-degrading bacterium, the type species for a new genus, Akkermansia, proposed in 2004 by Muriel Derrien and others.1,2,3,4,5,6,7, Extensive research is being undertaken to understand its association with obesity, diabetes, and inflammation. ... cranberries, pomegranate ..
Known contributor to a leaky Gut
- Acid-blocking medication (decreases B12, Zinc, )
- Antibiotics
- NSAIDS (non-steroid anti-inflammatories like asprin, ibuprofen, naproxen)
- Immune-suppressing
- medication (corticosteroids, methotrexate) (M -induces RA)
- Alcohol
- Excessive consumption of animal fats
- High fructose (fruit sugar) intake of corn syrup and agave nectar
- Infections (bacteria overgrowth, viral, parasitic, and yeast whose source of food is glucose)
- Stress (elevated cortisol levels)
- Environmental toxins
- PCBs
- Mercury
- Pesticides
- Cadium
1. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Xuan Zhang, Dongya Zhang, and Huijue et al. Jia. 2015. Nat Med 21, 8: 895–905.
2. The human microbiome: at the interface of health and disease. I. Cho and M.J. Blaser. 2012. Nature Reviews Genetics 13, 4: 260–270.
celiac-disease https://www.medicalhomeportal.org/living-with-child/diagnoses-and-conditions---faqs/celiac-disease
|
Jarisch-Herxheimer Reaction
A Herxheimer Reaction is a temporary worsening of symptoms following treatment for certain infections. Also know as a ‘Herx’ or a ‘Die-off’ reaction, it is caused by the release of endotoxins from bacteria that have been lysed by antibiotics or the immune system leading to additional inflammatory symptoms. This includes but is not limited to patients undergoing treatment for chronic Lyme disease and it’s Co-Infections (Bartonella, Babesia, Erlichia, Anaplasmosis, Mycoplasma). Underlying most symptoms of people with chronic Lyme disease is chronic inflammation, when a sudden release of endotoxin flares this inflammation; the result is a worsening of existing symptoms. The symptoms can be extremely debilitating and severe. For people experiencing this for the first time, it can be frightening and has lead to many late night trips to the hospital. Once people become familiar with their Herx symptoms, when to expect them, and how to manage them, it can actually be a welcome event as it signifies the treatment is working and progress is happening! |
|
|
Mycoplasma
- prokaryotic bacteria that lack cell wall Management during the acute phase may include antibiotics to eliminate the primary bacterial infection, especially if genitourinary, and NSAID to manage joint symptoms Treatments, similar to those used in psoriasis, may be required for cutaneous features Although the condition is usually self‐limiting, often resolving over a period of six months, it can progress to a chronic arthritis in around 15–20% of patients Aetiology Reactive Arthritis has been identified in people with a particular genetic type called HLA-B27. They have an increased chance of developing the syndrome - about 80% of people with reactive arthritis carry this gene Chlamydia trachomatis is the most commonly associated infection, although Neisseria gonorrhoea, salmonella, shigella, and campylobacter have all been associated Image: Reactive arthritis - palmoplantar pustulosis Reactive arthritis - psoriasiform hyperkeratosis Reactive arthritis - uveitis Video: https://www.youtube.com/watch?time_continue=114&v=S_v1oY2GZd8&feature=emb_title1 Source: 1. Mycoplasma Infections https://www.slideshare.net/doctorrao/mycoplasma 2. Increased risk of rheumatoid arthritis (RA) among patients with Mycoplasma pneumonia (MP): A nationwide population-based cohort study in Taiwan Kuo-An Chu, Weishan Chen, Chung Y. Hsu, et al. Published: January 14, 2019, https://doi.org/10.1371/journal.pone.0210750 This cohort study demonstrated that patients with MP had a higher risk of developing RA, especially in the first 2 years, in those aged younger than 19 and over 65. |
Increased risk of rheumatoid arthritis (RA) among patients with Mycoplasma pneumonia (MP): A nationwide population-based cohort study in Taiwan
Kuo-An Chu, Weishan Chen, Chung Y. Hsu, et al. Published: January 14, 2019, https://doi.org/10.1371/journal.pone.0210750
Kuo-An Chu, Weishan Chen, Chung Y. Hsu, et al. Published: January 14, 2019, https://doi.org/10.1371/journal.pone.0210750
- This cohort study demonstrated that patients with MP had a higher risk of developing RA, especially in the first 2 years, in those aged younger than 19 and over 65.
|
https://microbewiki.kenyon.edu/index.php/Mycoplasma_fermentans
M. ferementans is a pathogenic microbe that affects humans and is usually found in the genital area, and necrotizing tissue [9]. M. ferementans are being linked to diseases like rheumatoid arthritis (figure 2). Also studies suggest that M. ferementans causes chronic fatigue in patient [3]. Some of the strains studied like M. ferementans P.140 are known for causing severe respiratory infections and it has been demonstrated that the infection can travel through the blood to other organs such as the kidney, heart, and the brain [9]. It was discovered that during infection M. ferementans attack cells on the immune system (figure3), such as B cell, causing inflammatory reactions [4]. In humans it is being investigated that M. ferementans is able to enhance HIV replication, through the lipid-associated proteins present on their membrane [5]. M. ferementans seems to enhance the activation of Long term repeat gene, which is important during HIV infection, thorough a protein called toll receptor [5]. |
Enterovirus and enterovirus infects the gastrointestinal system these viruses are swallowed and they go into the mucosa and then they eventually end up in the lymph nodes where they're processed by the immune system and at some point they could become blood-borne and the theory is that when things go wrong and immunity doesn't happen when there's not cell normal cell me2day mediate immunity or antibody mediated immunity in the innate immunity system that that virus can end up in the central nervous system and that's when problems can occur and that's the theory.
References for linking a bad gut bacteria to some (not ciliac related) gluten and dairy intolerances are the proliferation of gluten and dairy-free foods and labeling in grocery stores. Something like 30% of the population is dairy-intolerant. This is in addition to the fact that ciproflocaxin can cured intolerances.
Can antibiotics such as ciprofloxacin trigger Autoimmune Diseases?
Ciprofloxacin is a fluoroquinolone (flor-o-KWIN-o-lone) antibiotic that fights bacteria in the body. Ciprofloxacin is used to treat different types of bacterial infections. It is also used to treat people who have been exposed to anthrax or certain types of plague.
Fluoroquinolone antibiotics can cause serious or disabling side effects. Ciprofloxacin should be used only for infections that cannot be treated with a safer antibiotic. https://www.drugs.com/ciprofloxacin.html
…...............................................................................................................................
Do they (Azithromycin, Ciprofloxacin) cause mitochondria damage resulting in Autoimune Diseases?
Can Antibiotics Trigger Autoimune Diseases ?
Can you recall prior to haveing an AD, taking an antibiotic?
Azithromycin is an antibiotic that fights bacteria. Azithromycin is used to treat many different types of infections caused by bacteria, such as respiratory infections, skin infections, ear infections, and sexually transmitted diseases.
To make sure azithromycin is safe for you, tell your doctor if you have:
Can antibiotics such as ciprofloxacin trigger Autoimmune Diseases?
Ciprofloxacin is a fluoroquinolone (flor-o-KWIN-o-lone) antibiotic that fights bacteria in the body. Ciprofloxacin is used to treat different types of bacterial infections. It is also used to treat people who have been exposed to anthrax or certain types of plague.
Fluoroquinolone antibiotics can cause serious or disabling side effects. Ciprofloxacin should be used only for infections that cannot be treated with a safer antibiotic. https://www.drugs.com/ciprofloxacin.html
…...............................................................................................................................
Do they (Azithromycin, Ciprofloxacin) cause mitochondria damage resulting in Autoimune Diseases?
Can Antibiotics Trigger Autoimune Diseases ?
Can you recall prior to haveing an AD, taking an antibiotic?
Azithromycin is an antibiotic that fights bacteria. Azithromycin is used to treat many different types of infections caused by bacteria, such as respiratory infections, skin infections, ear infections, and sexually transmitted diseases.
To make sure azithromycin is safe for you, tell your doctor if you have:
- liver disease, kidney disease, myasthenia gravis, a heart rhythm disorder; or a history of Long QT syndrome.
Patrice Carney in Belgium
What Is Leaky Gut?
Leaky gut syndrome may trigger or worsen such disorders as Rheumatoid Arthritis, Crohn's Disease, Celiac Disease, and Asthma.
What Is a Colic Baby?
Research at Guy's Hospital in 1999 linked one of the causes of colic to <http://en.wikipedia.org/wiki/Lactose_intolerance> lactose intolerance.</a>
Epidemiology suggests that chocolate, brassica, onions, and cow's milk are among the foods that a lactating mother may need to avoid.[3]
research, initially by Professor Kearney at Cork University Hospital (published 1995)[56] and then by Dr Dipak Kanabar at Guy's Hospital in London clearly demonstrated that infant colic was linked to transient lactose intolerance and could be controlled by pre-incubating the baby's feed with lactase enzyme. This is now the preferred method of treating colic in UK and Ireland using commercial lactase enzyme drops sold over-the-counter or on NHS prescription
only 5% of colic cases are caused by illness (most commonly by intestinal allergy, described above).[78]
78-Barr, RG (1998). "Colic and crying syndromes in infants". Pediatrics 102 (5 Suppl E): 1282–6. PMID 9794970.
They suggest treating the crying with daily doses of probiotics, or "good bacteria" (such as Lactobacillus acidophilus or Lactobacillus reuteri). In one study,[57] 83 colicky babies given Lactobacillus reuteri had reduced crying time. After one week, treated babies had close to 20% less crying time (159 min/day vs. 197 min/day). By 4 weeks, treated babies had 74% less crying (51 min/day vs. 197 min/day). Overall, there was a 95% positive response to the Lactobacillus reuteri probiotic drops in colicky infants.
Leaky gut syndrome may trigger or worsen such disorders as Rheumatoid Arthritis, Crohn's Disease, Celiac Disease, and Asthma.
What Is a Colic Baby?
Research at Guy's Hospital in 1999 linked one of the causes of colic to <http://en.wikipedia.org/wiki/Lactose_intolerance> lactose intolerance.</a>
Epidemiology suggests that chocolate, brassica, onions, and cow's milk are among the foods that a lactating mother may need to avoid.[3]
research, initially by Professor Kearney at Cork University Hospital (published 1995)[56] and then by Dr Dipak Kanabar at Guy's Hospital in London clearly demonstrated that infant colic was linked to transient lactose intolerance and could be controlled by pre-incubating the baby's feed with lactase enzyme. This is now the preferred method of treating colic in UK and Ireland using commercial lactase enzyme drops sold over-the-counter or on NHS prescription
only 5% of colic cases are caused by illness (most commonly by intestinal allergy, described above).[78]
78-Barr, RG (1998). "Colic and crying syndromes in infants". Pediatrics 102 (5 Suppl E): 1282–6. PMID 9794970.
They suggest treating the crying with daily doses of probiotics, or "good bacteria" (such as Lactobacillus acidophilus or Lactobacillus reuteri). In one study,[57] 83 colicky babies given Lactobacillus reuteri had reduced crying time. After one week, treated babies had close to 20% less crying time (159 min/day vs. 197 min/day). By 4 weeks, treated babies had 74% less crying (51 min/day vs. 197 min/day). Overall, there was a 95% positive response to the Lactobacillus reuteri probiotic drops in colicky infants.
|
Gut bacteria, specifically Prevotella histicola, have anti-inflammatory benefits that could help treat autoimmune disorders such as rheumatoid arthritis, lupus and ankylosing spondylitis. Researchers examined the possibility using mice, and more studies are planned. Mayo Clinic rheumatology chair Eric Matteson, M.D., explains why this is a hot area of research.
|
Sed ut perspiciatis unde omnis iste natus error sit voluptatem accusant doloremque laudantium, totam rem.
|
Catalyst - 'Gut Reaction' Part 1
|
Uncultured eubacterium WD2100 partial 16S rRNA gene, clone WD2100 WD2100 (1), (2)
uncultured eubacterium WD2100 partial 16S rRNA gene, clone WD2100GenBank: AJ292623.1
FASTA Graphics
LOCUS AJ292623 1500 bp DNA linear ENV 02-APR-2001 DEFINITION uncultured eubacterium WD2100 partial 16S rRNA gene, clone WD2100. ACCESSION AJ292623 VERSION AJ292623.1 KEYWORDS ENV; 16S ribosomal RNA; 16S rRNA gene. SOURCE uncultured eubacterium WD2100 ORGANISM uncultured eubacterium WD2100 Bacteria; environmental samples. REFERENCE 1 (bases 1 to 1500) AUTHORS Nogales,B., Moore,E.R., Llobet-Brossa,E., Rossello-Mora,R., Amann,R. and Timmis,K.N. TITLE Combined use of 16S ribosomal DNA and 16S rRNA to study the bacterial community of polychlorinated biphenyl-polluted soil JOURNAL Appl. Environ. Microbiol. 67 (4), 1874-1884 (2001) PUBMED 11282645 REFERENCE 2 (bases 1 to 1500) AUTHORS Nogales,B. TITLE Direct Submission JOURNAL Submitted (07-JUL-2000) Nogales B., Biological Sciences, University of Essex, Wivenhoe Park, Colchester, CO4 3SQ, UNITED KINGDOM FEATURES Location/Qualifiers source 1..1500 /organism="uncultured eubacterium WD2100" /mol_type="genomic DNA" /db_xref="taxon:133637"
LOCUS AJ292623 1500 bp DNA linear ENV 02-APR-2001 DEFINITION uncultured eubacterium WD2100 partial 16S rRNA gene, clone WD2100. ACCESSION AJ292623 VERSION AJ292623.1 KEYWORDS ENV; 16S ribosomal RNA; 16S rRNA gene. SOURCE uncultured eubacterium WD2100 ORGANISM uncultured eubacterium WD2100 Bacteria; environmental samples. REFERENCE 1 (bases 1 to 1500) AUTHORS Nogales,B., Moore,E.R., Llobet-Brossa,E., Rossello-Mora,R., Amann,R. and Timmis,K.N. TITLE Combined use of 16S ribosomal DNA and 16S rRNA to study the bacterial community of polychlorinated biphenyl-polluted soil JOURNAL Appl. Environ. Microbiol. 67 (4), 1874-1884 (2001) PUBMED 11282645 REFERENCE 2 (bases 1 to 1500) AUTHORS Nogales,B. TITLE Direct Submission JOURNAL Submitted (07-JUL-2000) Nogales B., Biological Sciences, University of Essex, Wivenhoe Park, Colchester, CO4 3SQ, UNITED KINGDOM FEATURES Location/Qualifiers source 1..1500 /organism="uncultured eubacterium WD2100" /mol_type="genomic DNA" /db_xref="taxon:133637"
The Bacterial, Archaeal and Plant Plastid Code
(transl_table 11) https://www.ncbi.nlm.nih.gov/Taxonomy/taxonomyhome.html/index.cgi?chapter=tgencodes#SG11
AAs = FFLLSSSSYY**CC*WLLLLPPPPHHQQRRRRIIIMTTTTNNKKSSRRVVVVAAAADDEEGGGG Starts = ---M------**--*----M------------MMMM---------------M------------ Base1 = TTTTTTTTTTTTTTTTCCCCCCCCCCCCCCCCAAAAAAAAAAAAAAAAGGGGGGGGGGGGGGGG Base2 = TTTTCCCCAAAAGGGGTTTTCCCCAAAAGGGGTTTTCCCCAAAAGGGGTTTTCCCCAAAAGGGG Base3 = TCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAG Click here to change format
Systematic Range and Comments: Table 11 is used for Bacteria, Archaea, prokaryotic viruses and chloroplast proteins. As in the standard code, initiation is most efficient at AUG. In addition, GUG and UUG starts are documented in Archaea and Bacteria (Kozak 1983, Fotheringham et al. 1986, Golderer et al. 1995, Nolling et al. 1995, Sazuka & Ohara 1996, Genser et al. 1998, Wang et al. 2003). In E. coli, UUG is estimated to serve as initiator for about 3% of the bacterium's proteins (Blattner et al. 1997). CUG is known to function as an initiator for one plasmid-encoded protein (RepA) in Escherichia coli (Spiers and Bergquist, 1992). In addition to the NUG initiations, in rare cases Bacteria can initiate translation from an AUU codon as e.g. in the case of poly(A) polymerase PcnB and the InfC gene that codes for translation initiation factor IF3 (Polard et al. 1991, Liveris et al. 1993, Sazuka & Ohara 1996, Binns & Masters 2002). The internal assignments are the same as in the standard code though UGA codes at low efficiency for Trp in Bacillus subtilis and, presumably, in Escherichia coli (Hatfiled and Diamond, 1993).
(transl_table 11) https://www.ncbi.nlm.nih.gov/Taxonomy/taxonomyhome.html/index.cgi?chapter=tgencodes#SG11
AAs = FFLLSSSSYY**CC*WLLLLPPPPHHQQRRRRIIIMTTTTNNKKSSRRVVVVAAAADDEEGGGG Starts = ---M------**--*----M------------MMMM---------------M------------ Base1 = TTTTTTTTTTTTTTTTCCCCCCCCCCCCCCCCAAAAAAAAAAAAAAAAGGGGGGGGGGGGGGGG Base2 = TTTTCCCCAAAAGGGGTTTTCCCCAAAAGGGGTTTTCCCCAAAAGGGGTTTTCCCCAAAAGGGG Base3 = TCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAGTCAG Click here to change format
Systematic Range and Comments: Table 11 is used for Bacteria, Archaea, prokaryotic viruses and chloroplast proteins. As in the standard code, initiation is most efficient at AUG. In addition, GUG and UUG starts are documented in Archaea and Bacteria (Kozak 1983, Fotheringham et al. 1986, Golderer et al. 1995, Nolling et al. 1995, Sazuka & Ohara 1996, Genser et al. 1998, Wang et al. 2003). In E. coli, UUG is estimated to serve as initiator for about 3% of the bacterium's proteins (Blattner et al. 1997). CUG is known to function as an initiator for one plasmid-encoded protein (RepA) in Escherichia coli (Spiers and Bergquist, 1992). In addition to the NUG initiations, in rare cases Bacteria can initiate translation from an AUU codon as e.g. in the case of poly(A) polymerase PcnB and the InfC gene that codes for translation initiation factor IF3 (Polard et al. 1991, Liveris et al. 1993, Sazuka & Ohara 1996, Binns & Masters 2002). The internal assignments are the same as in the standard code though UGA codes at low efficiency for Trp in Bacillus subtilis and, presumably, in Escherichia coli (Hatfiled and Diamond, 1993).