MMS - Miracle Mineral Supplement for Cancer
|
|
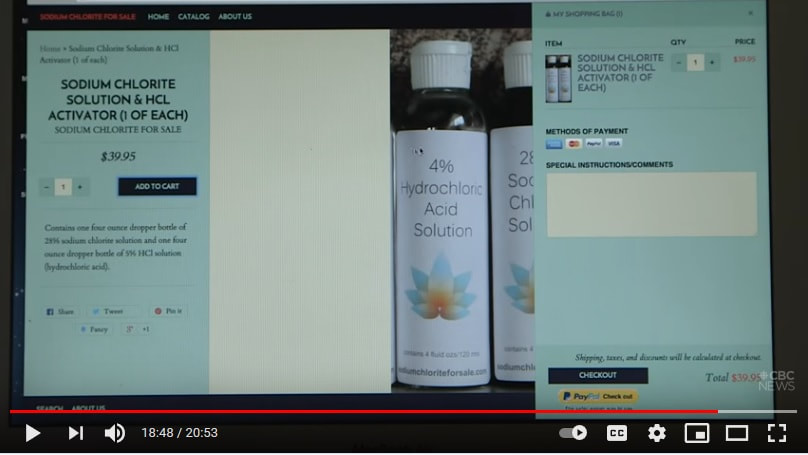
It is alleged this water purification drop could cure anything;
Cancer, Insomnia, Stomach Troubles, Diabetes, Autism ... MMS : The Miracle Cure? - the fifth estate https://www.youtube.com/watch?v=GpkxfPkmbUU Sodium chlorite, that's the active ingredient in MMS. The diluted form of bleach used to treat autism, is called CDS or chlorine dioxide solution. Two chemicals are mixed together to create chlorine dioxide, commonly used as an industrial bleach. Diluted with water, the believers claim MMS safely kills pathogens, cleansing the body. Health Canada says that's simply not true. Since 2010, the agency has issued seven public health warnings about MMS, repeating it is not safe for human consumption. |
|
Lynda Balneaves, leads the Centre for Integrative Medicine at the University of Toronto states: People that have consumed sodium chlorite have very serious side-effects including renal failure. They have had coagulation issues where they're bleeding out internally. It has life-threatening effects when it's consumed in an oral route. We also know it can be very corrosive and we see tissues being destroyed.
|
Acidified Sodium Chlorite (ASC) 2007 Chemical and Technical Assessment (CTA) Prepared by Madduri V. Rao, Ph.D.
for the 68th JECFA www.fao.org/fileadmin/templates/agns/pdf/jecfa/cta/68/Acidified_Sodium_Chlorite.pdf
for the 68th JECFA www.fao.org/fileadmin/templates/agns/pdf/jecfa/cta/68/Acidified_Sodium_Chlorite.pdf
The Food Industry has been using ASC for decades
ASC is intended for use as an antimicrobial agent as part of integrated, “multiple-hurdle” approach designed to control growth of microbial species. The ASC solution acts to reduce the number of pathogenic,
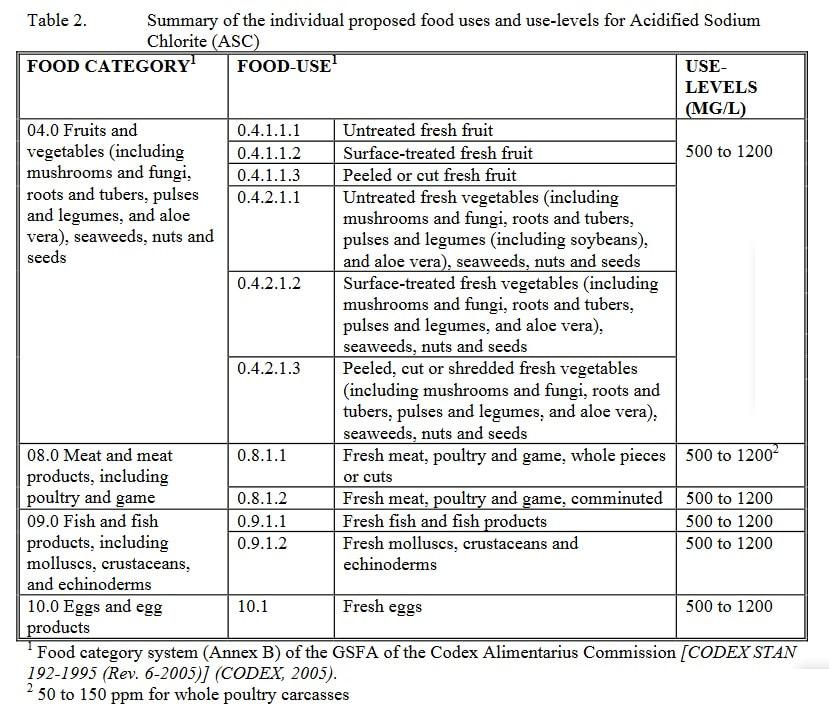
The solution is applied onto the surface of different types of fresh and processed foods (i.e., poultry meats, fresh and processed meats, fruits and vegetables, and freshwater fish and seafood) at relatively low levels (i.e., 50 to 1200 mg/l).
In most instances, residue levels declined to levels below the limits of detection with time.
- It exerts its antimicrobial activity by direct disruption of the cellular membrane, as well as by oxidation of cellular constituents
The solution is applied onto the surface of different types of fresh and processed foods (i.e., poultry meats, fresh and processed meats, fruits and vegetables, and freshwater fish and seafood) at relatively low levels (i.e., 50 to 1200 mg/l).
In most instances, residue levels declined to levels below the limits of detection with time.
Composition of sodium chlorite
The technical-grade of sodium chlorite used to prepare ASC is comprised of 80% of sodium chlorite, with sodium chloride, sodium carbonate, sodium hydroxide, sodium sulfate, and sodium chlorate making up the remainder of the composition.
Sodium chlorite [CAS No. 7758-19-2] is marketed in two forms, as a solid characterized by approx. 80% sodium chlorite and as an aqueous solution. Sodium chlorite is soluble in water, insoluble in non-polar solvents, and sparingly soluble in polar solvents (Merck, 2001).
Aqueous solutions are colourless to greenish yellow and exhibit a slight chlorine-like odour. While the chlorite ion is stable in aqueous solution, (depending in part on the pH of the solution, temperature, and alkalinity of the water) under acidic conditions, chlorite forms a semi-stable intermediate, chlorous acid (HClO2). Chlorous acid disintegrates to chlorine dioxide (ClO2), which further degrades to chlorite (ClO2 ̄) and ultimately chloride (Cl ̄) is formed.
Sodium chlorite [CAS No. 7758-19-2] is marketed in two forms, as a solid characterized by approx. 80% sodium chlorite and as an aqueous solution. Sodium chlorite is soluble in water, insoluble in non-polar solvents, and sparingly soluble in polar solvents (Merck, 2001).
Aqueous solutions are colourless to greenish yellow and exhibit a slight chlorine-like odour. While the chlorite ion is stable in aqueous solution, (depending in part on the pH of the solution, temperature, and alkalinity of the water) under acidic conditions, chlorite forms a semi-stable intermediate, chlorous acid (HClO2). Chlorous acid disintegrates to chlorine dioxide (ClO2), which further degrades to chlorite (ClO2 ̄) and ultimately chloride (Cl ̄) is formed.
6.1.2 Chlorine dioxide (ClO2)
Following acidification, a fresh ASC solution will typically contain less than 3 ppm of chlorine dioxide. Chlorine dioxide is relatively soluble and will therefore remain in solution; however, because chlorine dioxide also is extremely volatile, it will be off-gassed, with the off-gassing increasing as particle size decreases, from the solution when it is sprayed onto food. Likewise, for foods that are treated viaimmersion into the ASC solution, chlorine dioxide remaining in the solution is expected to evaporate from the food’s surface as the food dries. However, any chlorine dioxide that does come into contact with the food surface also may exert antimicrobial activity. Reaction of chlorine dioxide with microbial contaminants results in the formation of chlorite, which is further degraded to chlorate and, ultimately, chloride.
Following acidification, a fresh ASC solution will typically contain less than 3 ppm of chlorine dioxide. Chlorine dioxide is relatively soluble and will therefore remain in solution; however, because chlorine dioxide also is extremely volatile, it will be off-gassed, with the off-gassing increasing as particle size decreases, from the solution when it is sprayed onto food. Likewise, for foods that are treated viaimmersion into the ASC solution, chlorine dioxide remaining in the solution is expected to evaporate from the food’s surface as the food dries. However, any chlorine dioxide that does come into contact with the food surface also may exert antimicrobial activity. Reaction of chlorine dioxide with microbial contaminants results in the formation of chlorite, which is further degraded to chlorate and, ultimately, chloride.
6.1.3 Chloride (Cl ̄)
Although, chloride is the major by-product of the antimicrobial activity of the ASC solution, because of background sodium chloride levels in food, the chloride generated as a result of treatment with ASC solution is not quantifiable. However, given the ubiquitous nature of chloride ions in food, the amount of chloride produced as a by-product of the interaction of the oxy-chlorine species with microbial matter, is not expected to be a toxicological concern. Moreover, chloride occurs endogenously under normal physiological conditions in human body water.
Although, chloride is the major by-product of the antimicrobial activity of the ASC solution, because of background sodium chloride levels in food, the chloride generated as a result of treatment with ASC solution is not quantifiable. However, given the ubiquitous nature of chloride ions in food, the amount of chloride produced as a by-product of the interaction of the oxy-chlorine species with microbial matter, is not expected to be a toxicological concern. Moreover, chloride occurs endogenously under normal physiological conditions in human body water.
|
Antioxidant Activity, Bioactivities, Brazilian Fruitsarticle.sapub.org/pdf/10.5923.j.fph.20130301.05.pdf
human body, these actions reduce the incidence of cancer, inflammation, cataracts, macular degeneration and cardiovascular disease[ 3],[10-14]. Brazilian native fruits or those adapted to the tropical climate are surprisingly rich in bioactive compounds and have significant antioxidant activity[15-20]. Additionally, * Corresponding author: |
What happens if you mix sodium chlorite and citric acid ...https://www.quora.com/What-happens-if-you-mix-sodium-chlorite-and-citric-acid-together?share=1
Sodium Chlorite reacts with Citric Acid to form Chlorine Dioxide — a bleaching agent (i.e., bleach). Any over the counter drink that is advertised with to use such chemicals should be avoided at all costs. The FDA has received reports of severe vomiting and severe diarrhea which are linked to dehydration. |
MMS: The Miracle Mineral Supplement from Jim Humblehttps://www.earthclinic.com/supplements/mms.html
Initially used to treat malaria, the manufacturer claims field-tested success in treating and reversing the effects of AIDS, malaria, hepatitis, herpes, tuberculosis, most cancers, and a host of other diseases. |
|
Neutrophil-derived microvesicles enter cartilage and ...
https://stm.sciencemag.org/content/7/315/315ra190.full MVs originating from different cells differ in composition and biological function; for instance, in human RA, MVs of leukocyte, platelet, and synovial fibroblast origin can be found in synovial fluids, with concentrations positively correlating with disease activity (3, 4). Neutrophils accumulate in large numbers in the synovial space during ... |
Microvesicles (MVs) as biological targets for cancer diagnosis, prognosis, and therapy that enable MVs as new source and of new material and promising approach for practical therapeutics. |
Microvesicles Released from Human Red Blood Cells: Properties and Potential Applications
By Duc Bach Nguyen, Thi Bich Thuy Ly and Ingolf Bernhardt Submitted: October 30th 2016 Reviewed: May 5th 2017 Published: July 12th 2017
www.intechopen.com/books/novel-implications-of-exosomes-in-diagnosis-and-treatment-of-cancer-and-infectious-diseases/microvesicles-released-from-human-red-blood-cells-properties-and-potential-applications
By Duc Bach Nguyen, Thi Bich Thuy Ly and Ingolf Bernhardt Submitted: October 30th 2016 Reviewed: May 5th 2017 Published: July 12th 2017
www.intechopen.com/books/novel-implications-of-exosomes-in-diagnosis-and-treatment-of-cancer-and-infectious-diseases/microvesicles-released-from-human-red-blood-cells-properties-and-potential-applications
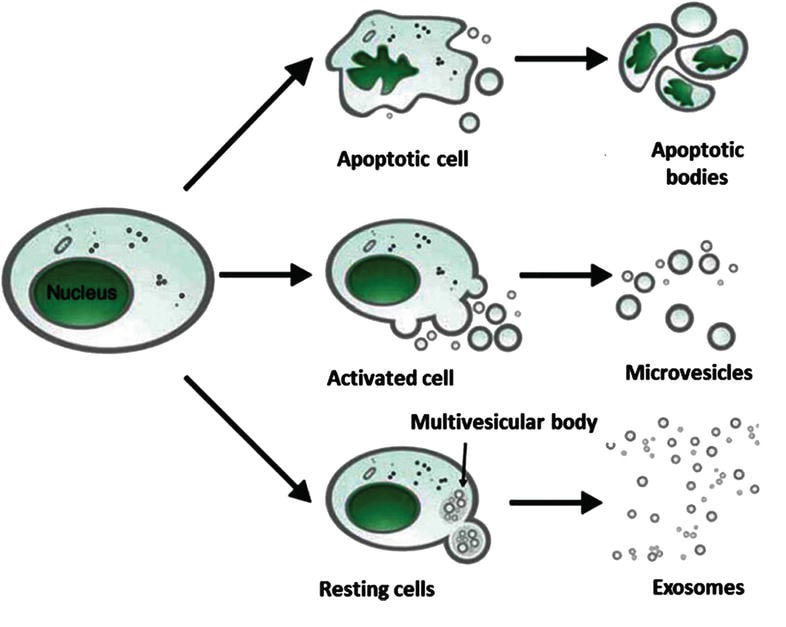
This chapter focuses on the formation and release of MVs from human RBCs and considers the isolation and characterization of MVs in order to apply MVs as potential vehicles for nucleic acid delivery. Similar to EVs released from nucleated cells, MVs from human RBCs carry biomarkers originated from plasma membrane and also microRNAs but not DNA. These properties suggest that MVs can be used as potential vehicles to transport proteins, nucleic acids, or signal molecules. While the understanding of the biogenesis of MVs in human RBCs and their physiological role remains limited, accumulating data suggest that MVs may be applied in cancer therapy. This chapter reviews our current knowledge pertaining to MVs released from human RBCs. It describes the formation and biological properties of MVs and mentions the potential application of MVs as a molecular vehicle for drug and nucleic acid delivery. Furthermore, it gives an introduction in the application of MVs for cancer treatment.
Although MVs have been discovered for years, the understanding of the mechanism of the formation as well as the biological roles of MVs is still a matter of debate. Recent reported findings led to advances of our understanding of the mechanism of formation and the role of MVs in many different diseases such as vascular diseases, cancer, infectious diseases, diabetes mellitus, diabetes, inflammation, and pathogen infection [24]. Inhibition of the production of MPs may serve as a novel therapeutic strategy for some diseases, especially for cancer treatment [11, 23, 26, 27]. In the next part of this chapter, the biogenesis, properties, and biological function of MVs released from human red blood cells (RBCs) are mainly addressed.
- EVs have been implicated in important biological processes such as surface-membrane trafficking and horizontal transfer of proteins and RNAs among neighboring cells, and distant tissues. Therefore, they play an important role in cell-to-cell communication under both physiological and disease conditions [11].
- These vesicle-associated complexes were capable of activating MHC class II leading to a restriction of T-cell responses. This finding suggests a role for exosomes in antigen presentation in vivo [32].
- That is the reason why MVs or exosomes are used in cancer diagnosis. For example, circulating levels of MVs are elevated in gastric cancer patients. In these patients, MPs released from CD41a-positive platelets are significantly increased in stage IV compared with stage I or II/III [35].
- The data suggest that MVs could be used as potential intracellular delivery vehicles for biomedical applications [44].
- Clinical studies have highlighted elevated levels of EVs in patients with severe malaria disease, and EVs have been linked to increased sequestration of infected RBCs to the endothelium [46].
- It has been demonstrated that reorganization or disruption of the cytoskeleton plays an important role in the release of MVs [36].
- By adding ascorbic acid to RBCs during storage, a significant decrease in MVs formation was observed [62].